|
12/9/2023 0 Comments High entropy alloys 
Meanwhile, only a few studies have been reported to clarify the catalytic and surface atomic-level structural relationships of HEA electrocatalysis probably because high-entropy factors increase so-called versatility, i.e., the structural and compositional complexities, such as atomic-level uniformity of elemental distribution and localised electronic properties. To date, various application studies have been reported for these HEA systems with respect to various types of electrocatalysis 14, 15, 16, 17, 18, 19, 20, 21. High-entropy alloys (HEAs), which generally comprise more than five elements, have recently attracted considerable attention because of their unique thermodynamical 12 and chemical properties 13. Therefore, atomic- and nano-level explorations of alloy catalyst surfaces that are directly correlated with the activity and durability of catalysts have always been a challenging issue for material development in practical electrocatalysts. atomic arrangements and alloy compositions in the surface vicinities, dynamically change due to power generations 1, 11. Furthermore, pristine and atomic-level surface structures, e.g. However, even though remarkable developments have been achieved in atomic-resolution transmission electron microscopy 9, 10, information for precise surface atomic structures even in Pt-based binary alloys remains insufficient because both the outermost- and near-surface of alloy catalysts are generally incredibly complicated even before use (in the pristine state). constituent elements 3, alloy compositions 4, crystallinity order 5, 6 and atomic arrangement 7, 8. Through these studies, broad consensus has been reached for high-performance catalyst surfaces relative to each of the various structural factors, e.g. For example, Pt-based binary alloys have been intensively studied 1, 2 because their catalytic properties determine the performance of polymer electrolyte membrane fuel cell (PEMFC) under harsh operating conditions such as strong acidity and severe potential fluctuations. 
The atomic-level surface designs of various alloys are key for improving the surface catalytic properties that are essential for development of novel electrocatalytic materials because alloy surfaces are structurally more complicated from the viewpoints of kinds of constituent elements and their compositions. The usefulness of the platform was demonstrated by showing the outperforming oxygen reduction reaction properties of high-entropy alloy surfaces when compared to Pt-Co binary surfaces. Nanometre-thick epitaxially stacking layers of Pt and equi-atomic-ratio Cr-Mn-Fe-Co-Ni, the so-called Cantor alloy, were synthesised on low-index single-crystal Pt substrates (Pt/Cr-Mn-Fe-Co-Ni/Pt(hkl )) as a Pt-based single-crystal alloy surface model for oxygen reduction reaction (ORR) electrocatalysis. 
The platform provides essential information that is crucial for the microstructural fundamentals of electrocatalysis, i.e., the detailed relationship between multi-component alloy surface microstructures and their catalytic properties. In this study, we propose an experimental study platform that enables the vacuum synthesis of atomic-level-controlled single-crystal high-entropy alloy surfaces and evaluates their catalytic properties. A comprehensive understanding of the relationship between surface atomic-level structures and catalytic properties is essential to boost the development of novel catalysts. 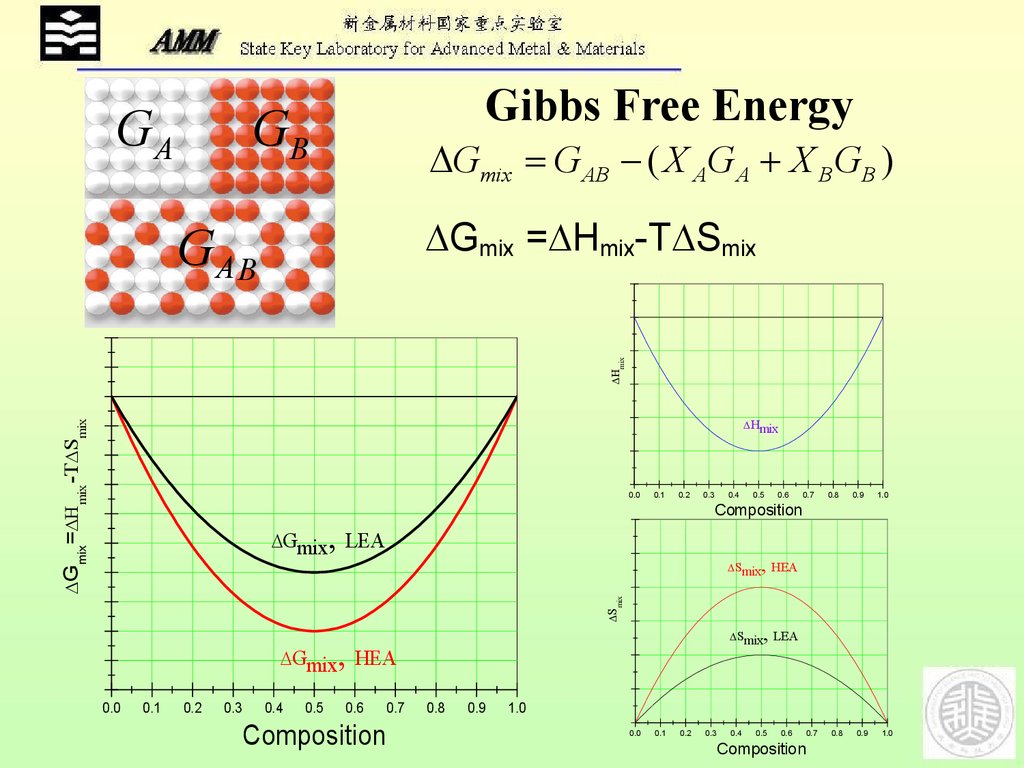
High-entropy alloys (HEAs) have attracted considerable attention to improve performance of various electrocatalyst materials. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
